[5/6/20 update]
Remdesivir
The big news since my last update was, of course, the NIH study of Remdesivir. Get the results straight from the horse’s mouth here. This isn’t a scientific paper, just a press release, but a press release from the NIH carries a lot more weight than a press release from a company with a financial incentive to overstate the results. This study was a randomized controlled trial – the gold standard – with 1,063 patients. Remdesivir is given intravenously, so the trial only included people who were already hospitalized and quite sick. People who got the drug recovered in a median of 11 days, compared to people who got the placebo and recovered in a median of 15 days. Fewer people died in the treatment group as well, but death is such a rare outcome, even for people hospitalized with COVID-19, that it takes a gigantic sample to be sure the effect is real. More trials are coming up, but this is already great news on its own. Two days after the NIH trial came out, the FDA issued an emergency use authorization for Remdesivir.
Two other Remdesivir trials happened to come out the same day, creating some confusion. Gilead (the maker of the drug) released its own fairly modest study comparing two courses of treatment: 5 days versus 10 days. The study doesn’t tell us much, but it wasn’t meant to. Last, a peer-reviewed study of Remdesivir in China came out in The Lancet. They found some benefit for a faster recovery, just like the US study, but they had to end their trial early because they ran out of COVID-19 patients. Their sample size ended up too small to give us much confidence in the result. But, together with the US study, we can be quite confident that Remdesivir causes people to recover from COVID-19 faster.
Hydroxychloroquine
This “rapid systematic review” from May 2nd found there were 7 completed trials of HCQ, with mixed results and generally low quality. That doesn’t mean HCQ is bad, just that we still don’t know enough to say one way or the other.
Favipiravir
I can’t find any new results since my last update. This NYT story quotes a doctor saying there are no trial results yet.
Convalescent Plasma
This great review from April 30th in the Journal of the American Medical Association summarizes what we know to date about convalescent plasma as a treatment for COVID-19. That is, taking blood from people who have recovered from COVID-19, doing some processing, and injecting the result into sick people. We still know very little. It’s not clear that this treatment even works well for other viruses. This summary says that prior studies on Ebola and the season flu found no benefit to convalescent plasma. The arm of the NIH that ran the remdesivir study has chosen not to run a study of convalescent plasma. Most of the treatments we read about in the news are not part of a clinical trial, so they won’t tell us much about how effective it is, if at all. There are, however, at least some small trials underway. They just haven’t finished yet.
[4/16/20 update]
HCQ
After my last post, a friend pointed me to a previous, small RCT of HCQ in patients with mild COVID-19 symptoms. It found no benefit.
A newly released RCT of 150 patients measured recovery every few days for four weeks and found no difference at any point between the treatment and control groups.
This review, dated April 14, says there are 142 ongoing trials of HCQ. Only five have been published, including the French observational study with no control group. Their summary is that the evidence does not yet support HCQ as a treatment for COVID-19.
FPV
This WSJ article says the only clinical trial to date is this one, which is a new draft of the FPV paper discussed below. It says six or seven others are ongoing.
Remdesivir
The stock price of Gilead, the manufacturer of Remdesivir has jumped on this very thin report that it might work. The trial has no control group and the report isn’t actual results, it’s just some recorded verbal comments that leaked from one of 152 (!) different clinical trial sites. Actual results are expected by the end of the month. I’d wait until then to draw any conclusions.
Convalescent Plasma
This article claims 200 people have been treated so far. Still waiting on clinical trial results.
[4/1/20 update]
HCQ
The first RCT of HCQ as a treatment for coronavirus came out today. It was a small but positive step. 62 patients with pneumonia that was described as “mild” were randomized to receive standard care alone or standard care with a daily dose of HCQ for five days. The patients who got HCQ got over their cough and fever about one day faster on average. CT scans showed somewhat larger improvement in their pneumonia as well. Four patients in the control group advanced to “severe illness” versus none in the group who received HCQ. This study was too small and too short to tell us if HCQ decreases the infection fatality rate. It also doesn’t tell us if people who take HCQ are less likely to contract coronavirus.
The French study that kicked off the excitement measured viral load, which this study did not. The mechanism could be that HCQ acts as an antiviral, but apparently it also modulates the immune system, so it might just help our bodies not overreact to the virus. Larger, longer studies in people at various stages of the disease should be forthcoming.
[3/29/20 updates]
HCQ
The French scientists who released an early paper with promising initial results for HCQ have released a new paper. The new paper is…not what I expected. The first one had a small treatment group and a small control group, but the group assignments weren’t random or blinded. We should cut them a lot of slack for throwing together a preliminary study as fast as possible. The logical next step would be a proper randomized controlled trial. Instead, they’ve gone in the opposite direction. The new paper had more patients, but didn’t have any control group at all. Out of 80 patients, 1 died. That’s right on the nose of the infection fatality rate of 1%. On the other hand patients were selected after they were hospitalized, so that result might be better than typical. Still, without a control group, this tells us almost nothing.
Still waiting on a number of larger, higher quality RCTs.
FPV
I can’t find any new published research.
Remdesivir
“Two trials in China are scheduled to end as soon as next month.”
Convalescent Plasma
I’ll add convalescent plasma – blood from people who have recovered from COVID – to the list of treatments I check up on regularly. Five critically ill patients were treated with convalescent plasma in an uncontrolled trial in China (published here). Randomized controlled trials should start treatments this week (see here).
[original post from 3/21/20]
I’m going to starting attempting to keep track of research into antiviral treatments for coronavirus. Unlike a vaccine, these could help if given to people who are already sick, meaning we wouldn’t need to produce nearly as much to make a difference. It’s possible but not certain they could also be used prophylactically.
I’ll ignore in vitro (petri dish) research because there is so much of it and so little of it translates to effective and practical treatments in humans. I’ll also ignore human anecdotes for the same reasons and because they’re often just not true.
Hydroxychloroquine (HCQ)
HCQ looks like one of the more promising treatments in the short-term. It’s been used for malaria for a long time and seems to be pretty safe when given to otherwhile healthy people. It’s possible it’s not as safe when given to people with COVID-19 and there’s a good chance it just doesn’t help them.
This half-decent French study published on March 17 suggests it probably does help clear the virus more quickly from people with moderate symptoms (hospitalized, but not in the ICU). The sample size was small and the control group wasn’t randomized or blinded. The researchers followed-up with the treatment group every day whereas they only got medical records from the control group. We can see that the treatment group was tested for the virus daily, whereas the control group was tested more sporadically. It’s possible there are other differences between the two control groups that we don’t know about. Still, patients in the treatment group appear to have been cleared of the virus much faster. The plot below shows the fraction of patients who tested positive for the virus by day since the beginning of treatment.
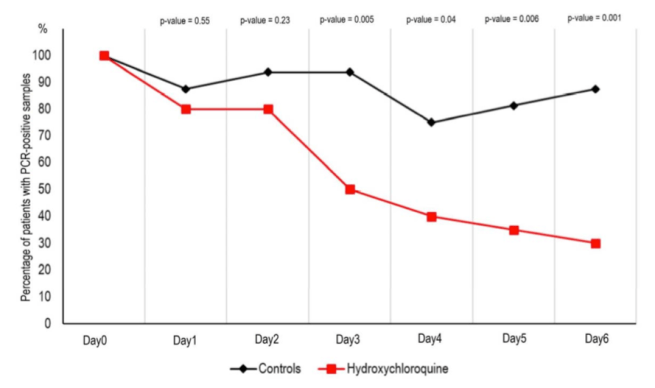
In this small trial, treatment started an average of 4 days after the onset of symptoms. The benefit could be greater if the treatment were given earlier (but that has to wait until there’s a positive test result). The authors propose that if further trials verify effectiveness of the treatment, the drug could be given as prevention. Without knowing how long it would take to scale up production, starting by giving this to healthcare workers could make a big difference, if it works.
This isn’t enough evidence to start a massive treatment program. It is enough to justify quickly running larger, higher quality clinical trials, which is exactly what’s happening.
Favipiravir (FPV)
Similar to HCQ, there is already an early trial of FPV, previously approved to treat flu. Like the other one, this trial was relatively small and non-randomized, but did include a group of people with different treatment as a control.
The lines in the plot below show what fraction of the patients test positive for the virus by day. ‘LPV’ is another drug used as control. As you can see, the treatment group seemed to clear out the virus faster. Separate results showed their lung function also improved faster.
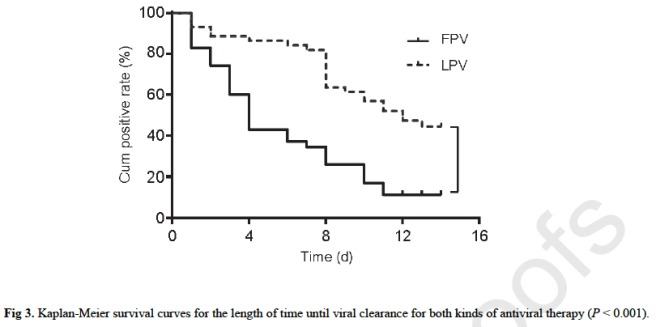
Further clinical trials are underway and apparently if everything goes very well FPV could be approved in May (in Britain, but presumably a similar timeline elsewhere).
Remdesivir
This drug has never been approved for anything, as far as I can tell, but has been tried as an antiviral several times in the past. Three of the first nine US COVID-19 patients were given remdesivir. They all recovered, but so do 99% of people, so that doesn’t mean anything. Several clinical trials are already underway, but there’s no strong indication one way or the other yet.

